A1.1 - Water (SL/HL)
Paper 1A – Multiple Choice
Question 1
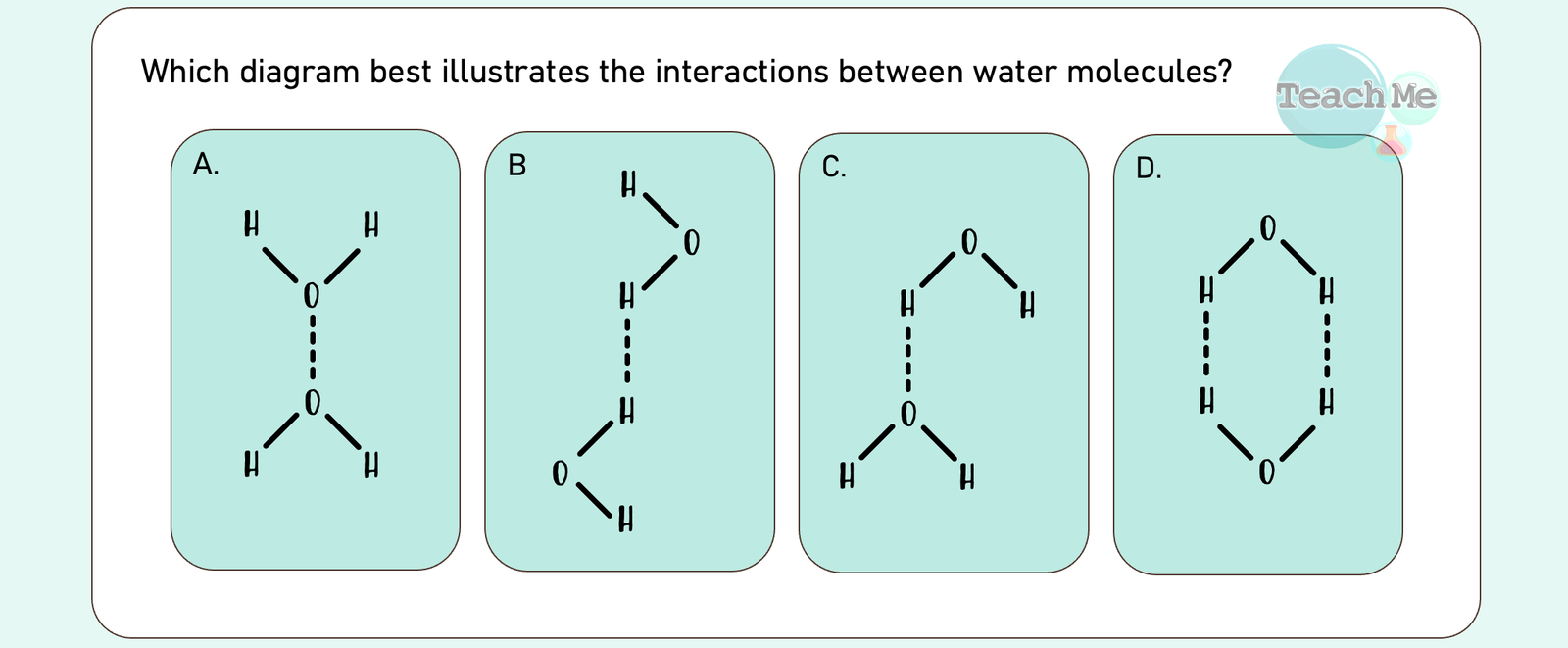
Reveal Big Brain Answer
Answer: C
🧠 Big Brain Explanation:



another. This is the only diagram that represents real intermolecular interactions in water 
❌ D | Shows two hydrogen bonds between the same pair of water molecules. Only one hydrogen bond can form between a pair at a time
Question 2

Reveal Big Brain Answer
Answer: A
🧠 Big Brain Explanation:
✅ A | Hydrogen bonding in water occurs between different water molecules, where the slightly negative oxygen (δ–) of one molecule is attracted to the slightly positive hydrogen (δ+) of another.💧✨.
❌ B | Says oxygen is slightly positive and hydrogen slightly negative — this reverses the actual charges. In reality: oxygen = δ–, hydrogen = δ+.
❌ C | Places the interaction within a single water molecule. Hydrogen bonding is between molecules, not inside one.
❌ D | Also describes a bond within the same molecule. Again: hydrogen bonds only form between molecules.
Question 3

Reveal Big Brain Answer
Answer: C
🧠 Big Brain Explanation:
❌ A | Water does form hydrogen bonds, and this ability comes directly from its polarity.
❌ B | Water’s polarity makes it an excellent solvent for polar compounds — like salts and sugars.
✅ C | Water’s polarity allows it to interact with other polar or charged substances — not non-polar ones. Non-polar substances (like oils) do not dissolve in water because they cannot form favourable interactions with water molecules. So C is the one that is NOT a result of water’s polarity ❌💧.
❌ D | High surface tension is caused by strong hydrogen bonding, which again depends on water’s polarity.
Question 4
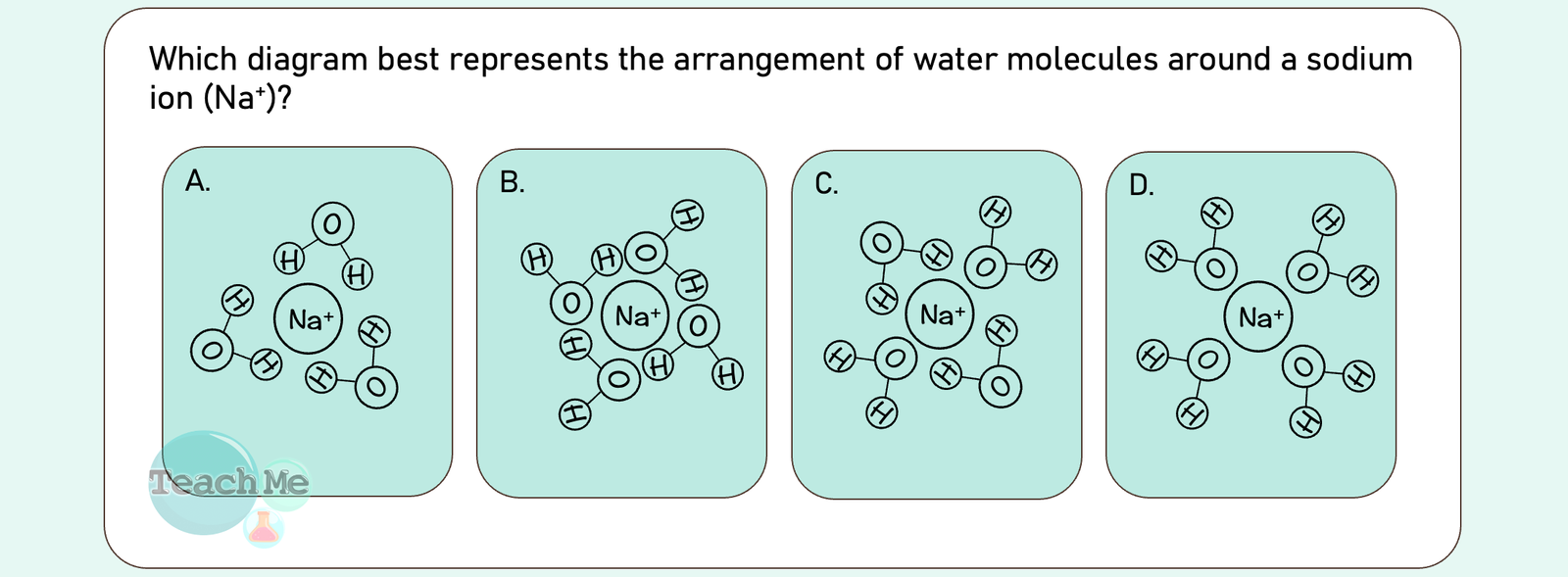
Reveal Big Brain Answer
Answer: D
🧠 Big Brain Explanation:
❌ A | The water molecules are pointing their hydrogen atoms (δ⁺) inward toward the positive ion. Positive-positive repulse each other (unstable).
❌ B |This shows a random mix of orientations. In reality, water molecules consistently align around an ion based on charge; they don’t just point any which way.
❌ C | Similar to A, this diagram incorrectly shows some hydrogen atoms (δ⁺) pointing toward the positive Na⁺ ion, which is not energetically favorable.
✅ D | Sodium ions are positively charged. Water is a polar molecule: the oxygen atom is slightly negative (δ⁻), and the hydrogen atoms are slightly positive (δ⁺). Opposites Attract: The slightly negative oxygen ends of the water molecules are strongly attracted to the positive sodium ion.
Question 5

Reveal Big Brain Answer
Answer: D
🧠 Big Brain Explanation:
❌ A | Small size and rapid movement relate to low viscosity, not high heat capacity.
❌ B | Ionization describes water’s role in pH, not its heat-absorbing ability.
❌ C | High kinetic energy means high temperature, not resistance to temperature change.
✅ D | Specific heat is the energy needed to raise temperature of a substance💧 Heating water doesn’t just make molecules move faster—it first has to break hydrogen bonds between them. In water a small temperature change requires absorption of a lot of heat energy. This property is referred to as having a high specific heat capacity.
Question 6

Reveal Big Brain Answer
Answer: C
🧠 Big Brain Explanation:
❌ A | This describes adhesion (water sticking to other polar substances).
❌ B | Water does not stick well to non-polar surfaces (it forms beads).
✅ C | Cohesion is the property of water where water molecules stick to other water molecules due to hydrogen bonding.
❌ D | Water molecules attract each other, not repel.
Question 7

Reveal Big Brain Answer
Answer: D
🧠 Big Brain Explanation:
❌ A | The lock-and-key model depends on enzyme shape (see C1.1), not water’s properties.
❌ B | Dissolving salt is due to water’s polarity (specifically its solvent property), not adhesion/cohesion.
❌ C | Cooling by sweating relies on water’s high heat of vaporization, not its adhesive/cohesive properties.
✅ D | Capillary action is the direct result of both properties working together: Adhesion: Water sticks to the walls of a narrow tube (e.g., xylem). Cohesion: Water molecules stick to each other, forming a continuous column. Together, they pull water upward against gravity. 🌱
Question 8

Reveal Big Brain Answer
Answer: B
🧠 Big Brain Explanation:
✅ I | Water forms hydrogen bonds with polar (charged) molecules. Thereby pulling them apart and dissolving them.
✅ II | Water is a polar molecule, meaning it has both (partial) positive & negative charges that attract polar molecules. It is this polarity that allows hydrogen bonds to form, thereby allowing water to dissolve substances.
❌ III | This property is about how well water transfers heat. It has nothing to do with dissolving solutes.
Since I, and II are correct the answer is B.
Question 9

Reveal Big Brain Answer
Answer: A
🧠 Big Brain Explanation:
✅ A | Water has a high latent heat of vaporisation, meaning it needs a lot of energy to turn from liquid into gas (vapour). When sweat evaporates from your skin, it uses up a lot of body heat, cooling you down.
This is the key reason water is an effective coolant in humans and other organisms.
❌ B | Cohesion makes water molecules stick to each other, not the skin. In addition, this doesn’t help explain cooling.
❌ C | Being a good solvent helps with transport of substances in the bloodstream, not with cooling.
❌ D | Buoyancy is about floating in water — completely unrelated to temperature regulation.
Question 10

Reveal Big Brain Answer
Answer: C
🧠 Big Brain Explanation:
❌ A | Condensation makes molecules bigger, not smaller.
❌ B | Breaking a disaccharide into two monosaccharides is hydrolysis, the opposite of condensation.
✅ C | A condensation reaction joins two molecules together, forming a larger molecule (in the process water is released). This is how disaccharides, triglycerides, and polypeptides are built in biology.
❌ D | Covalent bonds are formed in condensation, not hydrogen bonds. A different kind of covalent bond will form depending on the specific reaction. In the case of two amino acids joining together in a condensation reaction, the specific type of covalent bond formed is called a dipeptide bond.
Question 11

Reveal Big Brain Answer
Answer: B
🧠 Big Brain Explanation:
❌ A | That’s more related to water’s unique density, not its heat capacity.
✅ B | Water’s high specific heat means it resists temperature changes. This is vital for homeostasis, helping organisms (which are mostly water!) maintain a stable internal temperature despite external fluctuations. It also stabilizes temperatures in aquatic environments.
❌ C | Because of waters polarity is it able to have many properties, the ability to dissolve solutes (thanks to its polarity) is called the solvent property. The ability of water to dissolve solutes in unrelated to its high specific heat capacity.
❌ D | The opposite is true! Water’s high heat of vaporization means it resists evaporating, which is why sweating cools you so effectively.
Question 12

Reveal Big Brain Answer
Answer: C
🧠 Big Brain Explanation:
❌ I | These relate to density and temperature stability, not dissolving ability.
❌ II | Allows light penetration but doesn’t affect dissolving solutes.
✅ III | Waters ability to dissolve solutes is due to its polar nature and hydrogen bonding, which allows it to interact with and polar molecules (solvent property)
Only III directly explains why water is a good solvent. 💧✨
Question 13

Reveal Big Brain Answer
Answer: B
🧠 Big Brain Explanation:
✅ I | NaCl is polar, making it hydrophilic. Having a positively charged Na, and negatively charged Cl, it readily dissolves in water which is also polar.
❌ II | Lipids are non-polar (no positive or negative charges) making them hydrophobic, not hydrophilic. They therefor do not dissolve in water.
✅ III | Glucose is polar, making it hydrophilic, so it dissolves readily in water.
Therefore, only I and III are accurate.
Question 14

Reveal Big Brain Answer
Answer: C
🧠 Big Brain Explanation:
❌ A | Viscosity refers to the thickness/consistency of a substance. It affects flow/resistance, not upward force. Not the primary reason for floating.
❌ B | Oil is non-polar and therefor repels water. This keeps the bird dry & insulated despite being in water, but it doesn’t directly create buoyancy.
✅ C | Buoyancy (force of upthrust) is allows objects less dense than water to float. The bird’s body/air pockets provide lower density allowing it to float on water 🌊🦆
❌ D | Conductivity spreads heat from one place to another; it doesn’t make the bird float.
Question 15

Reveal Big Brain Answer
Answer: B





Question 16

Reveal Big Brain Answer
Answer: A
🧠 Big Brain Explanation:
Capillary action in xylem relies on two key properties: Adhesion: Water molecules are attracted to other surfaces (here, the hydrophilic xylem walls). Cohesion: Water molecules are attracted to each other via hydrogen bonds.
✅ A | Adhesion correct (attraction to walls). Cohesion correct (attraction to each other via H-bonds) 🌱💧
❌ B | Adhesion incorrect (describes dissolving ions, solvent property, not adhesion). Cohesion partially correct but describes “oxygen interactions” awkwardly; better to say H-bonds.
❌ C | Adhesion incorrect (describes cohesion instead). Cohesion incorrect (describes dissolving ions, solvent property, not cohesion).
❌ D | Adhesion incorrect (describes cohesion instead). Cohesion incorrect (describes adhesion instead).
Question 17

Reveal Big Brain Answer
Answer: C
🧠 Big Brain Explanation:
❌ I | This describes a phosphodiester bond (a kind of covalent bond) in the DNA backbone, not a hydrogen bond.
❌ II | This is a covalent bond within the water molecule (intramolecular), not a hydrogen bond, which occurs between two water molecules (intermolecular).
✅ III | Complementary DNA bases (A–T and G–C) are connected by hydrogen bonds.
Therefore, only III is accurate.
Question 18
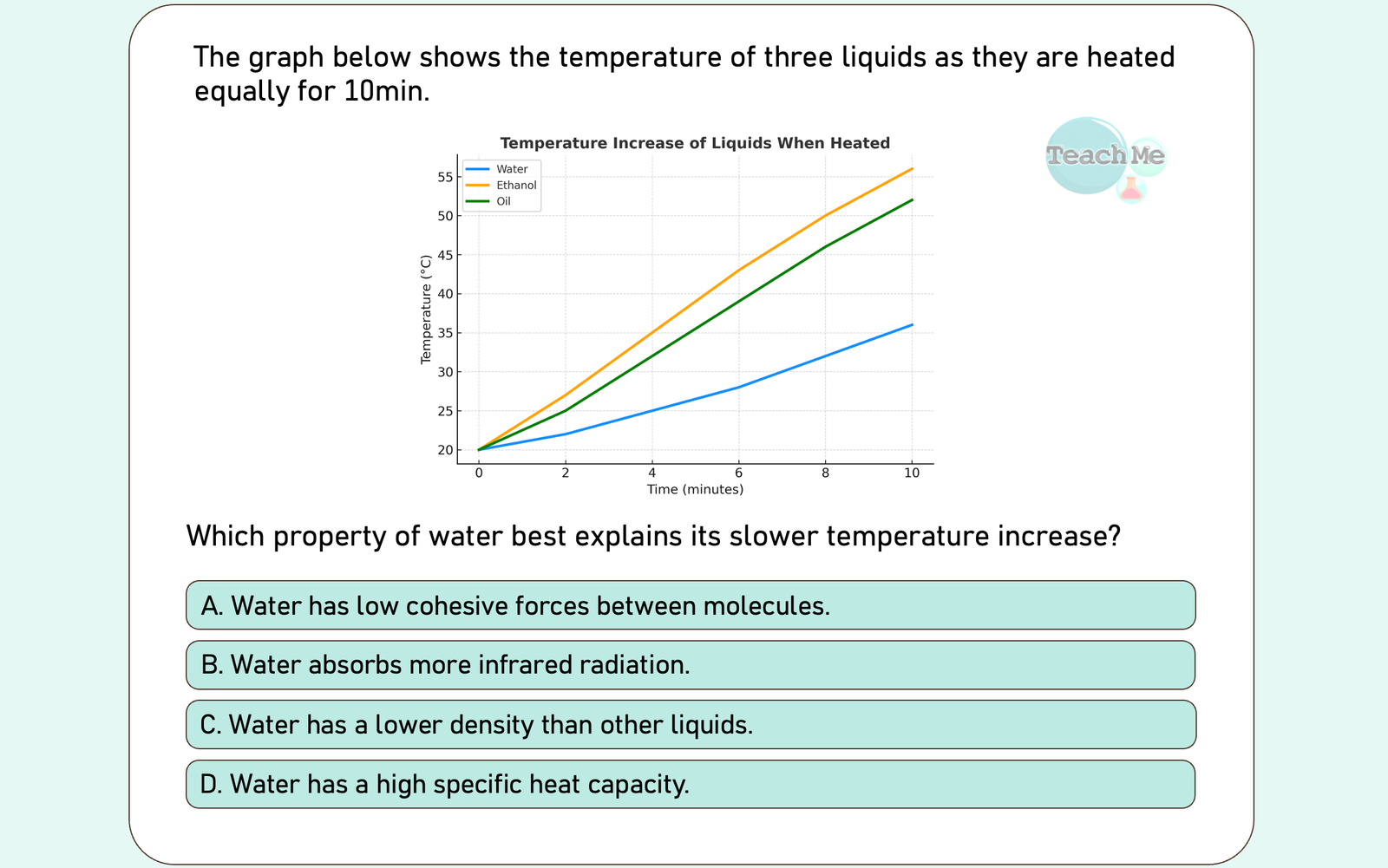
Reveal Big Brain Answer
Answer: D
🧠 Big Brain Explanation:
The graph shows water heats up slower than ethanol or oil when given the same heat energy.
❌ A | This relates to surface tension/adhesion, not heating rate.
❌ B | While true, this is not the standard property that explains slower temp increase under equal heating.
❌ C | Water’s density is unique but doesn’t directly explain its high resistance to temperature change.
✅ D | Water has a higher heat capacity which means it requires more energy to increase its temperature compared to ethanol or oil 🔥💧📈
Question 19

Reveal Big Brain Answer
Answer: A
🧠 Big Brain Explanation:
✅ A | Polarity occurs due to unequal sharing of electrons and is the key to hydrogen bonding, explaining cohesion, adhesion, solvent properties, and thermal regulation 💧⚡🔬
❌ B | O–H covalent bonds are strong. On the contrary, hydrogen bonds between molecules are weak allowing high fluidity.
❌ C | Each hydrogen atom in water can only form one hydrogen bond each, not two.
❌ D | Water is polar, which is why it dissolves hydrophilic substances, not hydrophobic ones.
Paper 2 – Short & Long Answer
Question 1

Reveal Big Brain Answer
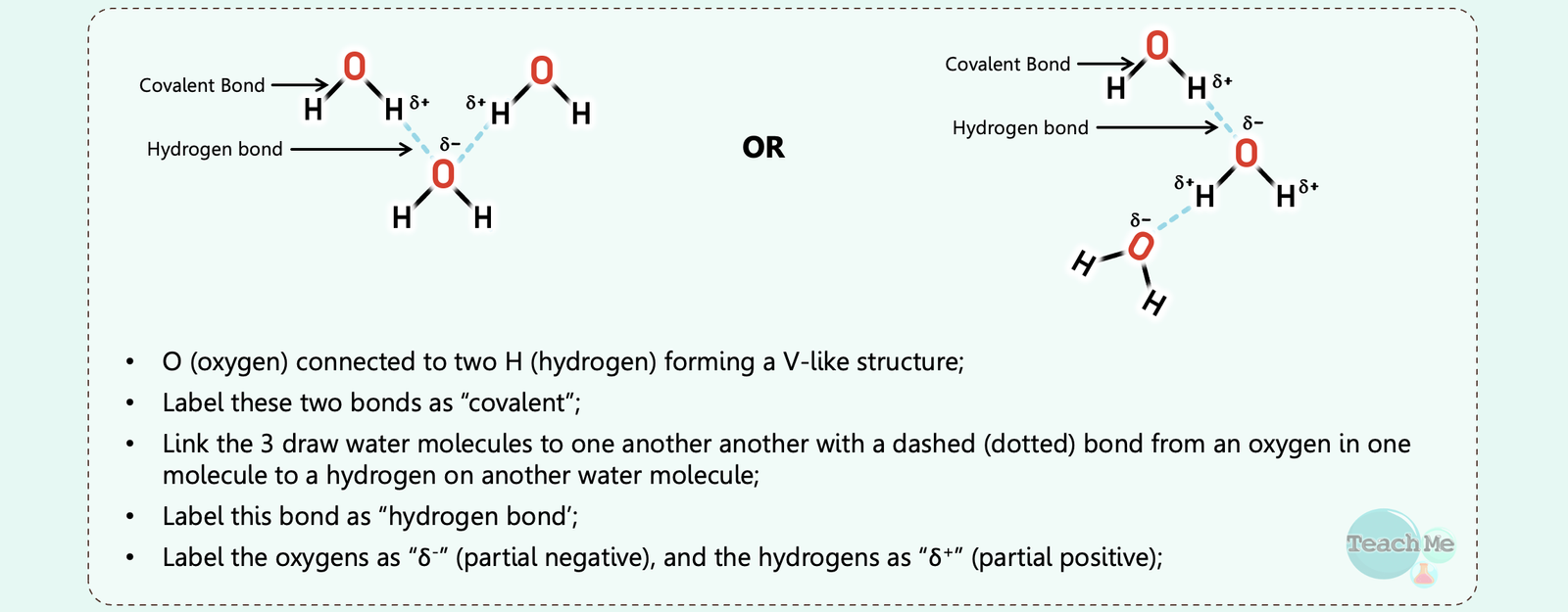
Question 2

Reveal Big Brain Answer

Question 3

Reveal Big Brain Answer

Question 4

Reveal Big Brain Answer

Question 5

Reveal Big Brain Answer





